Mitochondria – the organelles responsible for producing energy in human cells – were once free living organisms that found their way into the earliest eukaryotic cells more than a billion years ago. Since then, they have fused seamlessly with their hosts in a classic example of symbiotic evolution, and now rely on many proteins made in their host cell’s nucleus to function properly.
The outer membrane proteins of mitochondria are particularly important; they allow mitochondria to communicate with the rest of the cell and play a role in immune functions and a type of programmed cell death called apoptosis. During evolution, cells have developed a specific mechanism by which to insert these proteins – which are made in the cell’s cytoplasm – into the mitochondrial membrane. But what this mechanism was, and which cellular actors were involved, has long been a mystery.
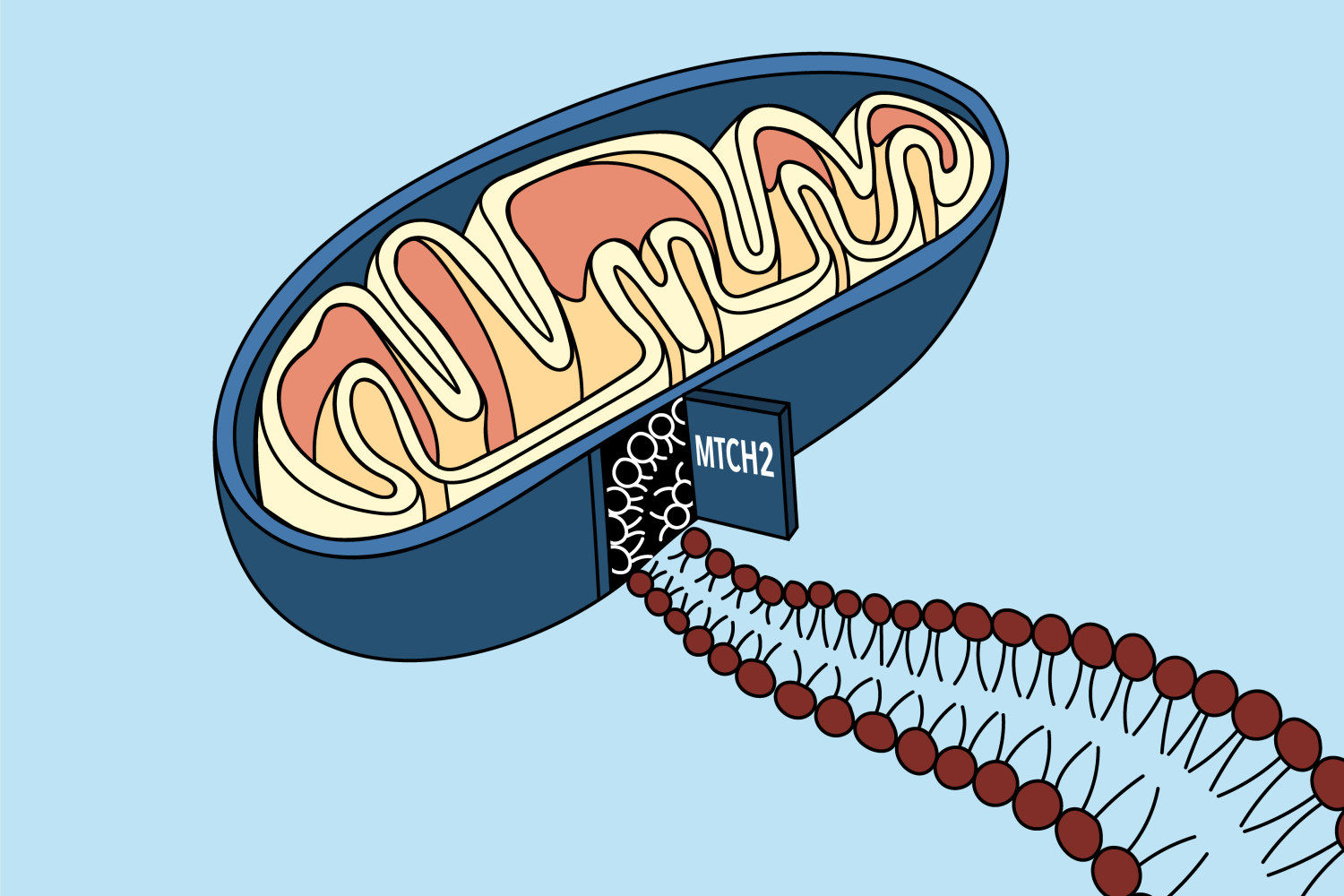
A new paper from the labs of MIT professor Jonathan Weissman and Caltech professor Rebecca Voorhees provides a solution to this mystery. The work, published on October 21 in the journal Sciencereveals that a protein called mitochondrial transporter homolog 2, or MTCH2 for short, which has been linked to many cellular processes and even diseases such as cancer and Alzheimer’s disease, is responsible for acting as a ” gate” allowing a variety of proteins to access the mitochondrial membrane.
“Until now, nobody knew what MTCH2 was really doing – they just knew that when you lose it, all these different things happen to the cell,” says Weissman, who is also a fellow at the Whitehead Institute for Biomedical Research and a researcher at the Howard Hughes Medical Institute. “It was somewhat of a mystery why this single protein affects so many different processes. This study provides a molecular basis for understanding why MTCH2 has been implicated in Alzheimer’s disease and lipid biosynthesis as well as fission and fusion mitochondrial: because it was responsible for inserting all these different types of proteins into the membrane.
“Collaboration between our labs has been key to understanding the biochemistry of this interaction and has led to a really exciting new understanding of a fundamental question in cell biology,” Voorhees said.
Finding a door
To uncover how proteins in the cytoplasm — specifically a class called tail-anchored proteins — were inserted into the outer membranes of mitochondria, Weismann Lab post-doctoral fellow and study first author Alina Guna, alongside Voorhees Lab graduate student Taylor Stevens and postdoc Alison Inglis, decided to use a technique called the CRISPR Interference Screening (or CRISPRi) approach, which was invented by Weissman and his collaborators.
“The CRISPR screen allowed us to systematically get rid of each gene and then watch and see what happened [to one specific tail-anchored protein]Guna says. “We found a gene, MTCH2, where when we got rid of it there was a huge decrease in the amount of our protein reaching the mitochondrial membrane. So we thought maybe that was the doorway to hall.
To confirm that MTCH2 was acting as a gateway into the mitochondrial membrane, the researchers performed additional experiments to observe what happened when MTCH2 was not present in the cell. They found that MTCH2 was both necessary and sufficient to allow tail-anchored membrane proteins to pass from the cytoplasm to the mitochondrial membrane.
The ability of MTCH2 to shuttle between proteins in the cytoplasm and the mitochondrial membrane is likely due to its specialized shape. The researchers ran the protein sequence through Alpha Fold, an artificial intelligence system that predicts a protein’s structure through its amino acid sequence, which revealed it was a protein. hydrophobic – perfect for inserting into the oily membrane – but with a single hydrophilic groove where other proteins might enter.
“It’s basically like a funnel,” says Guna. “Proteins come from the cytosol, they slip into this hydrophilic groove, and then move from the protein to the membrane.”
To confirm that this groove was important in the function of the protein, Guna and his colleagues designed another experiment. “We wanted to play with the structure to see if we could change its behavior, and we were able to do that,” says Guna. “We went in and did a point mutation, and that point mutation was enough to really change the behavior of the protein and its interaction with substrates. And then we went on and found mutations that made him less active and mutations that made him super active.
The new study has applications beyond answering a fundamental question in mitochondria research. “There are a lot of things that come out of it,” says Guna.
On the one hand, MTCH2 inserts proteins essential to a type of programmed cell death called apoptosis, which researchers could potentially exploit for cancer treatments. “We can make leukemic cells more susceptible to cancer treatment by giving them a mutation that alters the activity of MTCH2,” says Guna. “The mutation makes MTCH2 more ‘greedy’ and inserts more things into the membrane, and some of those things that have inserts are like pro-apoptotic factors, so those cells are more likely to die, which is fantastic in the context of cancer treatment.
The work also raises questions about how MTCH2 developed its function over time. MTCH2 evolved from a family of proteins called solute transporters, which transport a variety of substances across cell membranes. “We are really interested in this question of evolution: how do you evolve a new function from an old class of ubiquitous proteins? Weisman said.
And researchers still have a lot to learn about how mitochondria interact with the rest of the cell, including how they respond to stress and changes within the cell, and how proteins find their way to mitochondria first. venue. “I think that [this paper] is just the first step,” says Weissman. “This only applies to one class of membrane proteins – and it doesn’t tell you all of the steps that occur after the proteins are made in the cytoplasm. For example, how are they transported to the mitochondria? So stay tuned. listening – I think we’re going to learn that we now have a very good system to open up this fundamental element of cell biology.
#gate #mitochondrial #membrane