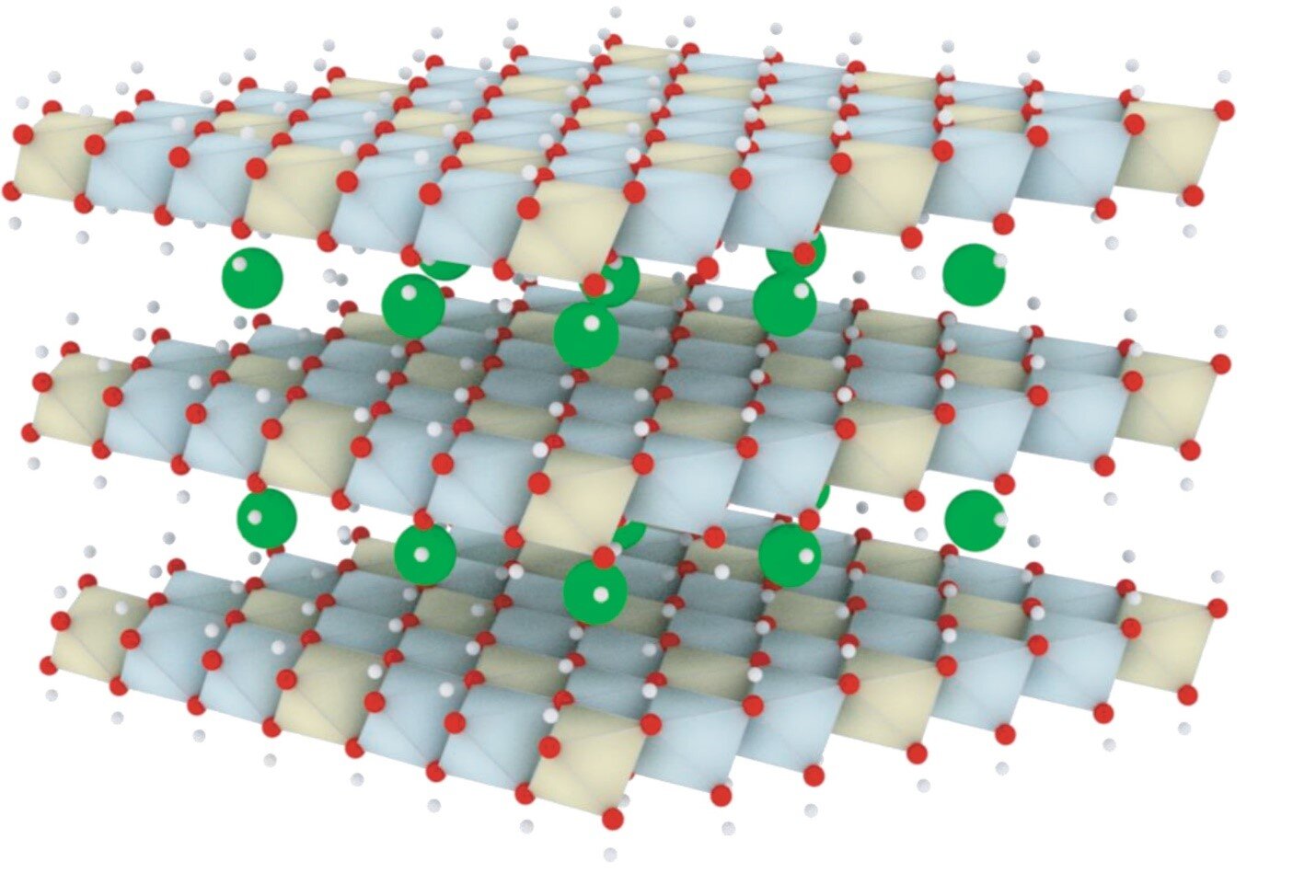
The idealized structure of LDH. Credit: Mingfei Shao, State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing
To guide the design and synthesis of electrocatalysts toward highly efficient oxygen evolution reactions (RELs), researchers from the Beijing University of Chemical Technology have summarized four common strategies for improving the REL performance of double hydroxides by layers (LDH) as well as the identification of active sites for LDH.
They published their work on September 7 in Advances in Energetic Materials.
“With the increasing demand and consumption of fossil fuels, energy shortage and environmental pollution are becoming severe and unavoidable,” said corresponding author Mingfei Shao, a professor at the State Key Laboratory of Energy Engineering. Chemical Resources of Beijing University of Chemical Technology, Beijing. “It is necessary to explore sustainable and renewable energies. Hydrogen, in particular, is a new energy with splendid application prospects.”
The production of highly pure hydrogen can be obtained by electrochemical separation of water using electricity transformed from renewable energies such as wind and solar. But like one of the half-reactions, the OER is a four-electron process, with low-efficiency energy use, according to Shao.
Shao and his team focus on LDH, a large-type two-dimensional material. The wide tunability, molar ratios and interlayer anions make it an exceptional catalyst for OER in alkaline media.
“We have summarized four common strategies applied to improve OER performance of LDH. Through these strategies, OER overpotential can be decreased, leading to high energy utilization efficiency,” Shao said. “Some work on the identification of active sites for LDHs is introduced. Revealing the reaction mechanism and active sites provides the theoretical guidance for designing efficient electrocatalysts.”
The development and exploration of OER catalysts are mostly in the experimental stage at present, which cannot meet the standards of large-scale practical use. For example, the problems of scaling up the size of catalysts and maintaining stability during OER remain. Moreover, most of the reported preparation methods of LDH-based catalysts are complicated and time-consuming, leading to high costs and limiting their application, according to Shao.
“The recognition of reactive oxygen species such as oxygen species adsorbed by active sites on the surface of electrocatalysts and the oxygen radical dispersed in solution during OERs still remains ambiguous due to the unstable and unstable existence apparent reactive oxygen species,” Shao said. “Having recognized these reactive oxygen species, how to leverage them for more effective RELs is always vital.”
“We hope this review can offer insights to further identify LDH active sites with the goal of providing guidance for designing more advanced electrocatalysts toward electrochemical water splitting,” Shao said.
Researchers build bifunctional catalysts for oxygen reduction and evolution reaction
Xin Wan et al, Layered Double Hydroxides for Oxygen Evolution Reaction to Efficient Hydrogen Generation, Advances in Energetic Materials (2022). DOI: 10.34133/2022/9842610
Provided by Beijing Institute of Technology Press
Quote: Layered Double Hydroxides for Oxygen Evolution Reactions (2022, October 26) Retrieved October 26, 2022 from https://phys.org/news/2022-10-layered-hydroxides-oxygen-evolution-reactions.html
This document is subject to copyright. Except for fair use for purposes of private study or research, no part may be reproduced without written permission. The content is provided for information only.
#Layered #double #hydroxides #oxygen #evolution #reactions