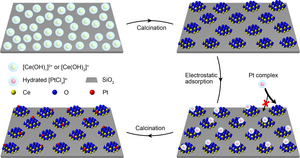
image: Processes for manufacturing functional cerium nanoglue and ceria-supported single Pt catalysts.
see After
Credit: Team of Prof. ZENG Jie
In a study published in Nature, a research team led by Prof. ZENG Jie of the University of Science and Technology of China (USTC) and international collaborators developed a new “nanoglue” strategy to stabilize atomically dispersed metal catalysts .
In the field of heterogeneous catalysis, the atomic dispersion metal catalyst has attracted considerable attention due to its unique geometric and electronic properties, highest atomic efficiency and uniform active sites. However, highly dispersed metal atoms easily move and clump together due to high surface energy, resulting in low stability, or strongly interact with the support and become catalytically passivated. Therefore, how to obtain “mobile but non-agglomerating” metal sites that can improve both catalytic activity and stability has always been a difficult point in catalysis.
Considering this, the research team designed a new type of “nanoisland” catalyst (also called nanoglue), in which the active metal atoms are isolated on “islands” where they can move respectively, but the migration towards neighboring “islands” is suppressed, thus obtaining “mobile but non-agglomerating” atomic sites.
To achieve this goal, suitable materials must be chosen for the “nanoislands” and the supports, respectively. The affinity between the metal atoms and the “nanoislands” should be much stronger than that between the metal atoms and the support. Otherwise, the metal atoms are easy to leave their own “nanoislands”. Therefore, researchers chose oxides with high affinity for metal atoms as “islands” (such as cerium oxide) in the designed model catalyst, and oxides with low interaction (such as silica) as support for stabilize the “islands”.
To effectively isolate the metal atoms, the “islands” must be of small enough size and high enough number density on the medium. Conventional synthetic methods (such as the impregnation method) tend to produce large, non-uniform particles that are unsuitable as “islands”. The team therefore developed a strong electrostatic adsorption method in an aqueous solution. The high-density cerium atoms were first coated on the surface of the silica, then they agglomerated into isolated “islands” of a size less than 2 nanometers by calcination, as shown in the figure below.
The next challenge is to precisely place the metal atoms on the “nanoislands” rather than on the support. To this end, the researchers again used the strong electrostatic adsorption method, accompanied by the point difference of zero charges of ceria and silica, to the oppositely charged ceria island and silica surface. The negatively charged platinum precursor could only be adsorbed onto the positively charged cerium oxide nanolands, rather than the negatively charged silica support, so the platinum atoms were grown exclusively on the islet of cerium oxide. Due to the limited surface area of the ultra-small “nanoislands” and the low concentration of platinum precursor, less than one platinum atom was deposited on each “island” on average.
The study showed that the platinum atoms on the cerium oxide “nanoislands” can resist sintering when calcined in air up to 600℃. In particular, platinum atoms have been shown to move to their own “island” under hydrogen reduction conditions at high temperature, instead of diffusing to the neighboring “island”. In addition, the carbon monoxide oxidation activity of the hydrogen-activated catalyst was increased by two orders of magnitude as well as exceptional stability.
This work provides a new strategy to boost both catalytic activity and stability. In the future, it is planned to apply this concept of “nanoglues” to different catalytic reactions by choosing appropriate supports, “nanolands” and active metal atoms.
This work was conducted in collaboration with WANG Yong from Washington State University, Bruce C. Gates from the University of California, Davis, and LIU Jingyue from Arizona State University.
The title of the article
Functional CeOx Nanoglues for Robust Atomic Dispersion Catalysts
Publication date of articles
26-Oct-2022
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of press releases posted on EurekAlert! by contributing institutions or for the use of any information through the EurekAlert system.
#Nanoglues #stabilize #metal #atoms #catalysis