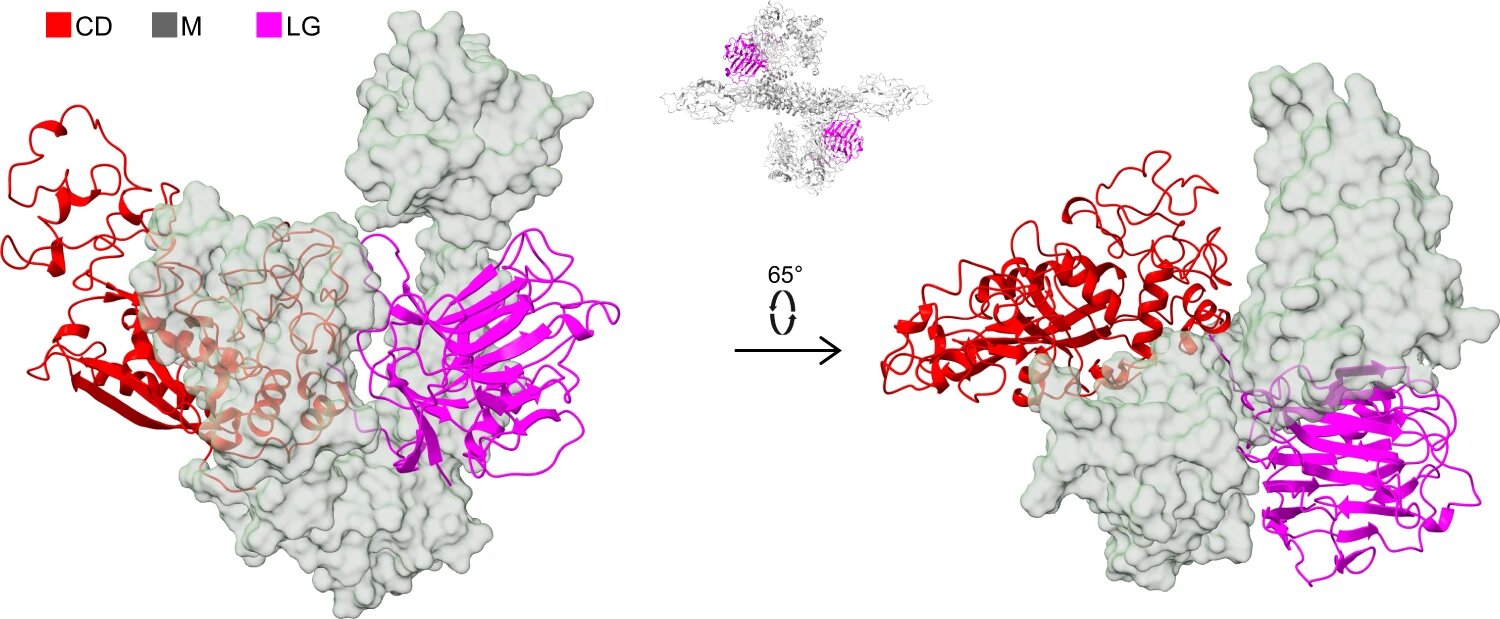
The laminin G (LG)-like domain of the PAPP-A subunit. Representation to emphasize the separation of the CD domain (red, cartoon) and the LG domain (magenta, cartoon) by the M domains (transparent surface) in two different orientations. The position of the LG domain in the PAPP-A·STC2 tetrameric structure is indicated by the inset above the arrow. Credit: Nature Communication (2022). DOI: 10.1038/s41467-022-33698-8
The laminin G (LG)-like domain of the PAPP-A subunit. Representation to emphasize the separation of the CD domain (red, cartoon) and the LG domain (magenta, cartoon) by the M domains (transparent surface) in two different orientations. The position of the LG domain in the PAPP-A·STC2 tetrameric structure is indicated by the inset above the arrow. Credit: Nature Communication (2022). DOI: 10.1038/s41467-022-33698-8Danish researchers have determined the three-dimensional structure of the proteolytic enzyme PAPP-A. The results may allow us to better understand the basic biology that regulates linear growth in vertebrates. The same regulatory mechanisms are also implicated in several age-related diseases, and so the research is an important step towards the development of new types of drugs.
The growth factor IGF plays a key role in human growth. In the absence of IGF signaling, we become dwarfs. Later in life, IGF is implicated in age-related diseases, such as cancer and cardiovascular disease. In either case, IGF must be converted from an inactive form to an active form. This is what PAPP-A is able to do.
“Seven years ago, we discovered that the STC2 protein blocks the activity of PAPP-A, thus indirectly inhibiting the activity of the growth factor IGF. To block the activity, STC2 must form a complex with PAPP-A We have studied this complex, and now we know its three-dimensional structure,” explains Professor Claus Oxvig.
“It’s fascinating to see what a molecule, which we know very well biochemically, actually looks like. PAPP-A is heart-shaped with an inner ‘chamber’. But from a research perspective, the shape is not The most interesting characteristic is rather the interactions between the different elements of the molecule.”
There are still many unanswered questions about the molecular mechanisms that regulate the amount of IGF converted to the active form. It is likely that complex formation between PAPP-A and STC2 is highly regulated. Such a hypothesis is supported by previous findings showing that natural human variants of STC2, in which only one amino acid is substituted, form the complex with PAPP-A slightly more slowly. The consequence is that slightly more IGF can be activated by PAPP-A, resulting in an increase in height of up to 2.1 cm.
The first author of the publication reporting the PAPP-A STC2 structure, graduate student Sara Dam Kobberø, used cryo-electron microscopy (cryo-EM) to determine the structure of the large protein complex. The Danish National Research Infrastructure Cryo-EM (EMBION, AU) enabled this study, which also involved participants from the University of Copenhagen.
The research has been published in Nature Communication.
More information:
Sara Dam Kobberø et al, Structure of proteolytic enzyme PAPP-A with endogenous inhibitor stanniocalcin-2 reveals its inhibitory mechanism, Nature Communication (2022). DOI: 10.1038/s41467-022-33698-8
Provided by Aarhus University
Quote: Researchers determine the three-dimensional structure of PAPP-A (2022, October 31) Retrieved October 31, 2022 from https://phys.org/news/2022-10-three-dimensional-papp-a.html
This document is subject to copyright. Except for fair use for purposes of private study or research, no part may be reproduced without written permission. The content is provided for information only.
#Researchers #determine #threedimensional #structure #PAPPA