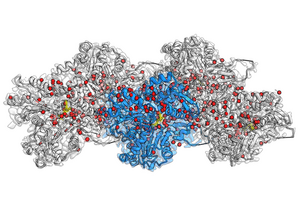
image: Cryo-EM reconstruction of F-actin bound to Mg2+-ADP-BeF3. at a resolution of 2.2 Å. The central actin subunit is colored blue, the other four subunits are gray. The densities corresponding to water molecules are colored in red and ADP in yellow.
see After
Credit: Molecular Physiology MPI
“We answer fundamental questions of life that scientists have been trying to answer for several decades,” Raunser remarks. In eukaryotic cells, actin proteins are abundant and tend to aggregate (polymerize) into filaments. These filaments make up the network that makes up the cytoskeleton of the cell and controls various cellular processes through movement. Immune cells, for example, use actin filaments to move and hunt bacteria and viruses. Researchers already knew that filament dynamics are regulated by ATP hydrolysis – the reaction of ATP with water that cleaves a phosphate group and generates energy. What previously remained unanswered, however, were the exact molecular details behind this process.
Too soft, too big? – not for cryo-EM
As actin filaments are too flexible or too large for X-ray crystallization and nuclear magnetic resonance, cryo-EM was the only viable technique to obtain detailed images. In 2015, Raunser’s team used cryo-EM to imagine a new three-dimensional atomic model of the filaments, with a resolution of 0.37 nanometers. In 2018, his group described the three different states that actin proteins acquire in the filament: bound to ATP, bound to ADP in the presence of the cleaved phosphate, bound to ADP after release of the phosphate.
How water molecules move
In their current study, Raunser and his colleagues were able to set a new resolution record: they obtained all three actin states with a resolution of around 0.2 nanometers, making previously invisible details visible. Three-dimensional maps not only display all amino acid side chains of proteins, but also reveal where hundreds of water molecules are placed. Thanks to the comparison between these new structures and those of isolated actin, they were able to deduce how water molecules move. During polymerization, water molecules move into the ATP pocket in such a way that only a single water molecule remains in front of the ATP, ready to attack a phosphate and initiate hydrolysis . The precision achieved with this approach can help further research in the field: “Our high-resolution model can propel scientists into designing small molecules for tissue light microscopy research, and ultimately into therapeutic applications” , says Raunser.
A door opener!?
The authors also shed light on the final fate of phosphate. Previously, scientists thought there was a back door in the ATP pocket that remains open after ATP hydrolysis to facilitate the exit of phosphate. However, the new cryo-EM structures show no trace of open backdoors. Therefore, the release mechanism remains a mystery. “We think there’s a door, but it’s probably opening momentarily,” comments Raunser, who now wants to use mathematical simulations and time-resolved cryo-EM methods to demonstrate how the phosphate comes out. Clearly, these exciting findings have opened the door for scientists to dig deeper in hopes of uncovering even more details behind the processes by which actin filaments contribute to cell movement.
Research method
Experimental study
Research subject
cells
The title of the article
Structural basis of actin filament assembly and aging
Publication date of articles
26-Oct-2022
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of press releases posted on EurekAlert! by contributing institutions or for the use of any information through the EurekAlert system.
#pocket #full #water #molecules #actin #filaments #drive #cell #movement