To preserve life on earth, effective purification methods are needed that can separate impurities from water and air. Therefore, for a long time, carbonaceous materials have been used to deodorize, separate and remove dangerous anionic contaminations by adsorption.
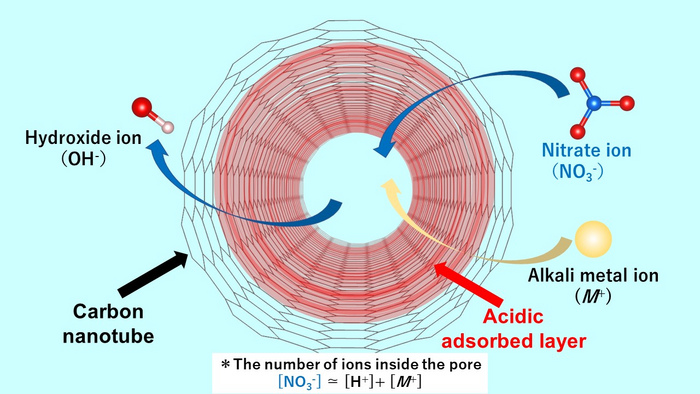
 The acidic adsorbed layer enhances the nanoconfinement of nitrate anion impurities in single-walled carbon nanotubes (SWCNTs) due to the strong confinement by the pore and the strong interaction between the layer and the anion. When nitrate ions are adsorbed, hydroxide ions are desorbed from the nanospace. Thus, the aqueous solution exhibits alkaline properties. Image Credit: Takahiro Ohkubo of Okayama University Department of Chemistry, Graduate School of Natural Science and Technology, Okayama University.
The acidic adsorbed layer enhances the nanoconfinement of nitrate anion impurities in single-walled carbon nanotubes (SWCNTs) due to the strong confinement by the pore and the strong interaction between the layer and the anion. When nitrate ions are adsorbed, hydroxide ions are desorbed from the nanospace. Thus, the aqueous solution exhibits alkaline properties. Image Credit: Takahiro Ohkubo of Okayama University Department of Chemistry, Graduate School of Natural Science and Technology, Okayama University.
Until now, the complete mechanism underlying how carbon purifies water has remained a mystery. Moreover, it is not established whether the aqueous solution adsorbed on the carbonaceous material is alkaline, acidic or neutral.
To address these shortcomings, scientists led by Dr. Takahiro Ohkubo, Associate Professor in the Department of Chemistry, Faculty of Natural Sciences and Technology, University of Okayama, Japan, examined the central mechanism by which carbon nanopores adsorb anions.
The scientists mention how they used Raman spectroscopic tools to analyze the adsorption of nitrate ions by the cylindrical pore of single-walled carbon nanotubes (SWCNTs).
Details of the research were first posted online on September 16e2022, then published in the Journal of Colloid and Interface Science (volume 629 part B).
Dr. Ohkubo and his contemporaries succeeded in decoding the mechanism of creation of the acid layer near the walls of the pores. It turns out that when an aqueous solution containing ions penetrates the carbonaceous material, even if the aqueous solution is neutral, an acidic aqueous layer is created containing protons which maintains a stable state.
To date, there have been no reports demonstrating the existence of acid adsorption layers formed within nanotubes of carbonaceous materials.
Dr. Takahiro Ohkubo, Associate Professor, Department of Chemistry, Faculty of Natural Science and Technology, Okayama University
Dr. Nobuyuki Takeyasu, member of the research team and associate professor in the same faculty of Okayama University, found that the acidic layer allows excellent adsorption of contaminations by negatively charged nitrate anions, where the amount adsorbed of nitrate ions is much larger than that of cations, or positively charged groups.
Additionally, hydroxide ions are produced as counterions. The anions in the bulk solution are exchanged with the hydroxide ions in the SWCNT, making the aqueous solution alkaline.
Researchers analyzed anion adsorption using many alkali metal nitrates, including solutions of rubidium nitrate, lithium nitrate, sodium nitrate, and cesium nitrate. They learned that compared to metal ions, more nitrate ions are adsorbed. The amount of proton adsorption was almost the same regardless of the type of alkali metal ion used.
The acidic layer in the pore can strongly adsorb nitrate anion species due to both the strong confinement by the pore and the strong interaction between the layer and the anion.
Dr. Takahiro Ohkubo, Associate Professor, Department of Chemistry, Faculty of Natural Science and Technology, Okayama University
These discoveries are crucial steps towards the engineering and development of carbon nanotubes ideal for ion adsorption and air and water decontamination. The purification mechanism formulated in this study is a unique model that elucidates the alkalinity of the aqueous solution medium, which was a mystery until now.
The scientists note that the results of their study strongly underscore the need to neutralize water before use when carbonaceous materials capture ionic impurities.
Another important contribution of this research is the illustration that the interface of nanomaterials is a unique chemical reaction field, which could guide further experiments. Based on the positive contributions, this study takes the understanding of the anion adsorption mechanism by carbon to the next level, paving the way for unique carbon nanotubes as effective purifiers.
Journal reference:
Ohkubo, T. et al. (2022) Nanoconfinement of anions reinforced by an acid layer in a cylindrical pore of single-walled carbon nanotube. Journal of Colloid and Interface Science. doi.org/10.1016/j.jcis.2022.09.070.
Source: http://www.okayama-u.ac.jp/index_e.html
#acid #layer #carbon #nanotubes #confinement #anionic #impurities